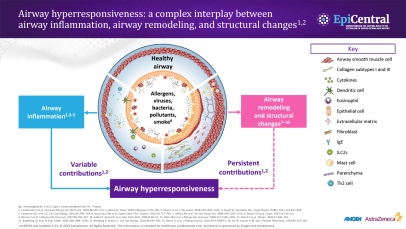
References
1. Chapman DG, Irvin CG. Clin Exp Allergy. 2015;45:706–719;
2. Comberiati P, et al. Immunol Allergy Clin North Am. 2018;38:545–571;
3. Borak J, Lefkowitz RY. Occup Med (Lond). 2016;66:95–105;
4. Berair R, et al. J Allergy (Cairo). 2013;2013:185971;
5. Allakhverdi Z, et al. J Allergy Clin Immunol. 2009;123:958–960;
6. Gunst SJ, Panettieri RA. J Appl Physiol (1985). 2012;113:837–839;
7. Porsbjerg CM, et al. Eur Respir J. 2020;56:2000260;
8. Busse WW. Chest. 2010;138(Suppl 2):4S–10S;
9. Crimi E, et al. Am J Respir Crit Care Med. 1998;157:4–9;
10. Jeffery PK, et al. Am Rev Respir Dis. 1989;140:1745–1753;
11. Boulet L-P, et al. Chest. 1997;112:45–52;
12. Booms P, et al. J Allergy Clin Immunol. 1997;99:330–337;
13. Gelb AF, Zamel N. Curr Opin Pulm Med. 2002;8:50–53;
14. Slats AM, et al. J Allergy Clin Immunol. 2008;121:1196–1202;
15. Ward C, et al. Thorax. 2002;57:309–316;
16. Heijink IH, et al. Allergy. 2020;75:1902–1917;
17. Fehrenbach H, et al. Cell Tissue Res. 2017;367:551–569;
18. Hough KP, et al. Front Med (Lausanne). 2020;7:191;
19. Gil FR, Lauzon A-M. Can J Physiol Pharmacol. 2007;85:133–140;
20. Bradding P. Eur Respir J. 2007;29:827–830;
21. Pelaia C, et al. J Clin Med. 2023;12:3371;
22. Raby KL, et al. Front Immunol. 2023;14:1201658;
23. Hollins F, et al. J Immunol. 2008;181:2772–2780;
24. John AE, et al. J Immunol. 2009;183:4682–4692;
25. Kaur D, et al. J Immunol. 2010;185:6105–6114;
26. Moiseeva EP, et al. PLoS One. 2013;8:e61579;
27. Kaur D, et al. Chest. 2012;142:76–85;
28. Suto W, et al. Int J Mol Sci. 2018;19:3036;
29. Robinson DS. J Allergy Clin Immunol. 2004;114:58–65;
30. Brightling CE, et al. N Engl J Med. 2002;346:1699–1705;
31. Kaur D, et al. Allergy. 2015;70:556–567;
32. Moir LM, et al. J Allergy Clin Immunol. 2008;121:1034–1039;
33. Woodman L, et al. J Immunol. 2008;181:5001–5007;
34. Tatler AL, et al. J Immunol. 2011;187:6094–6107;
35. Saunders R, et al. J Allergy Clin Immunol. 2009;123:376–384;
36. Saunders R, et al. Clin Transl Immunology. 2020;9:e1205;
37. Singh SR, et al. Allergy. 2014;69:1189–1197;
38. Begueret H, et al. Thorax. 2007;62:8–15;
39. Siddiqui S, et al. J Allergy Clin Immunol. 2008;122:335–341;
40. Bonvini SJ, et al. Eur Respir J. 2020;56:1901458;
41. Lai Y, et al. J Allergy Clin Immunol. 2014;133:1448–1455;
42. Altman MC, et al. J Clin Invest. 2019;129:4979–4991;
43. Al-Shaikhly T, et al. Eur Respir J. 2022;60:2101865;
44. Rayees S, Din I. Asthma: pathophysiology, herbal and modern therapeutic interventions. 1st ed. Cham, Switzerland: Springer International Publishing; 2021;
45. in‘t Veen JC, et al. Am J Respir Crit Care Med. 1999;160:93–99;
46. Leuppi JD, et al. Am J Respir Crit Care Med. 2001;163:406–412;
47. Rijcken B, Weiss ST. Am J Respir Crit Care Med. 1996;154:S246–S249;
48. Sears MR, et al. N Engl J Med. 2003;349:1414–1422;
49. Cockcroft DW, et al. Clin Allergy. 1977;7:235–243;
50. Boulet LP, et al. J Allergy Clin Immunol. 1983;71:399–406;
51. Reddel HK, et al. Eur Respir J. 2000;16:226–235;
52. Coates AL, et al. Eur Respir J. 2017;49:1601526;
53. Fowler SJ, et al. Am J Respir Crit Care Med. 2000;162:1318–1322;
54. Vandenplas O, et al. Eur Respir J. 2014;43:1573–1587;
55. Weiler JM, et al. J Allergy Clin Immunol. 2016;138:1292–1295;
56. O’Byrne PM, Inman MD. Chest. 2003;123:411S–416S;
57. Hallstrand TS, et al. Eur Respir J. 2018;52:1801033.