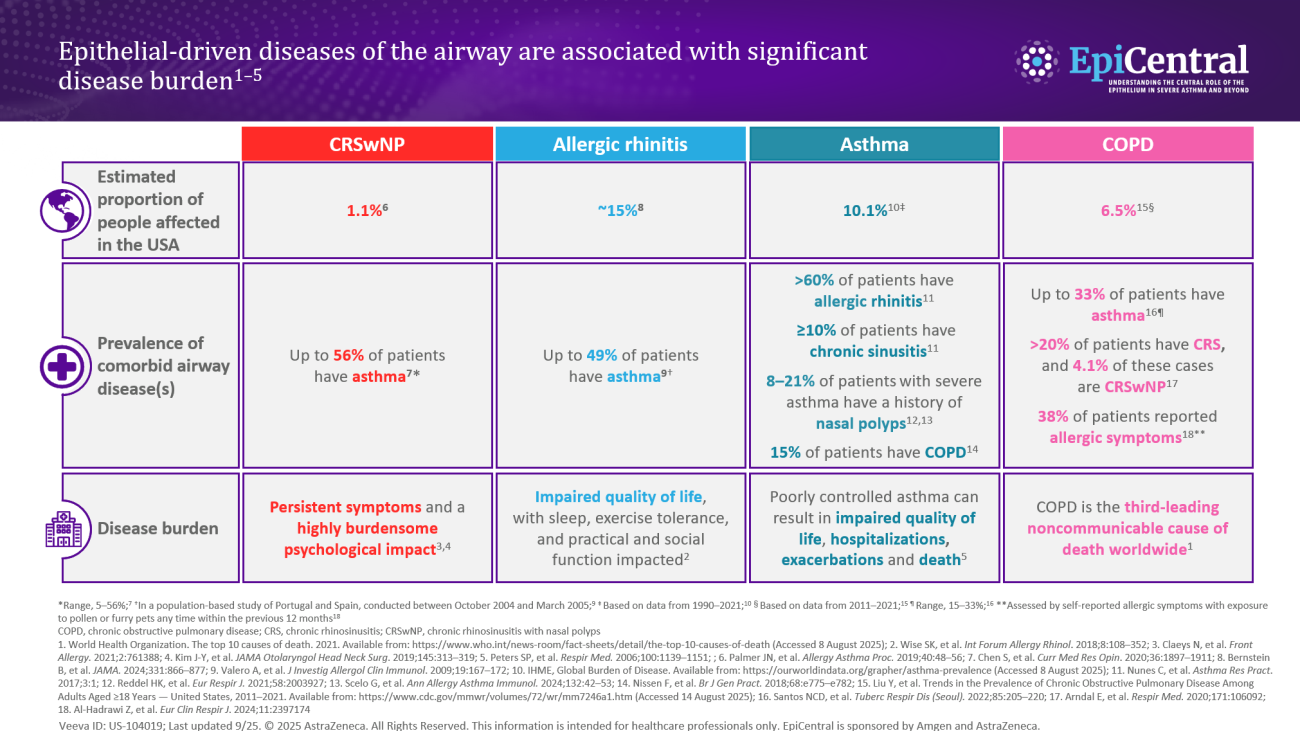
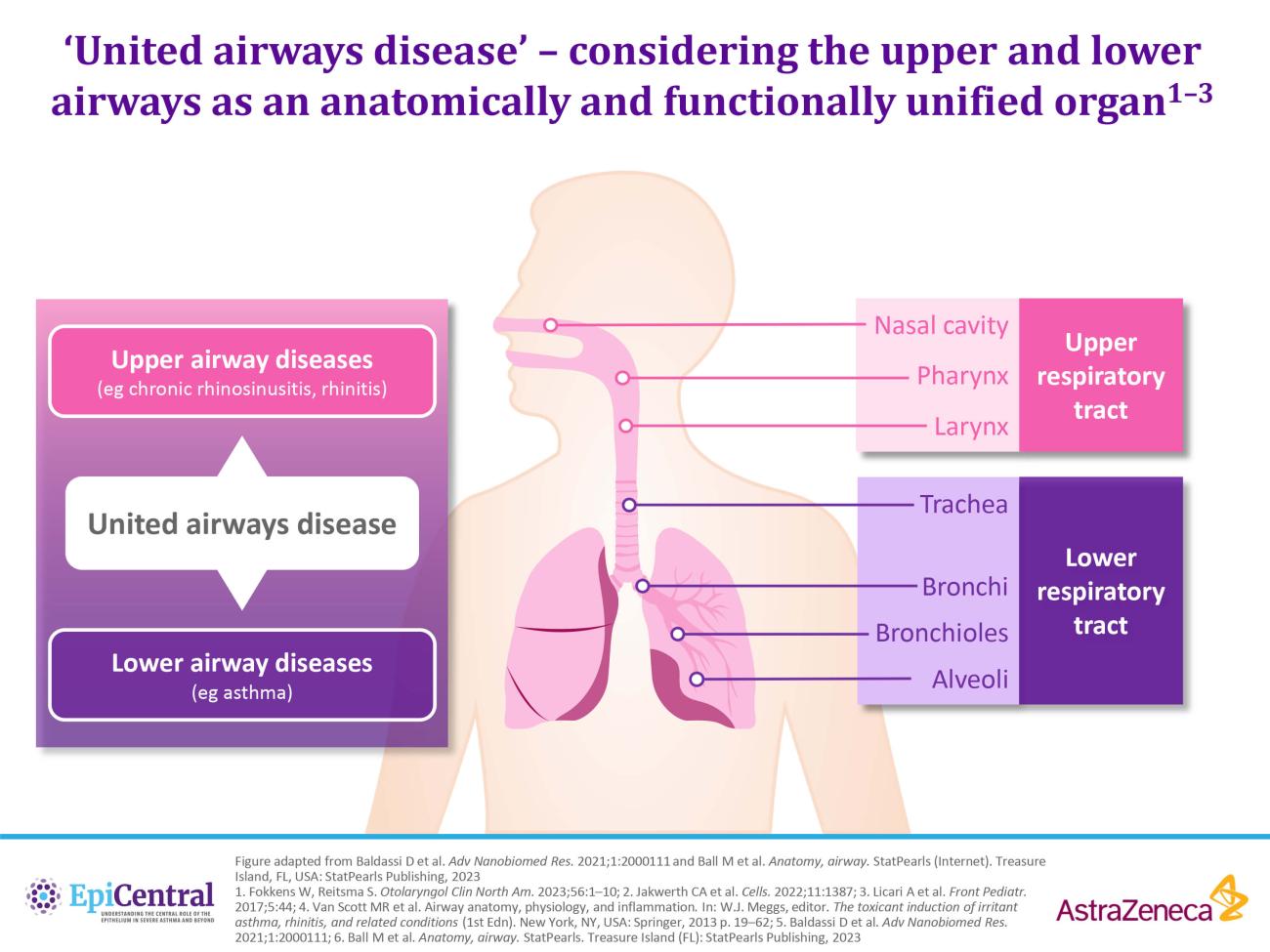
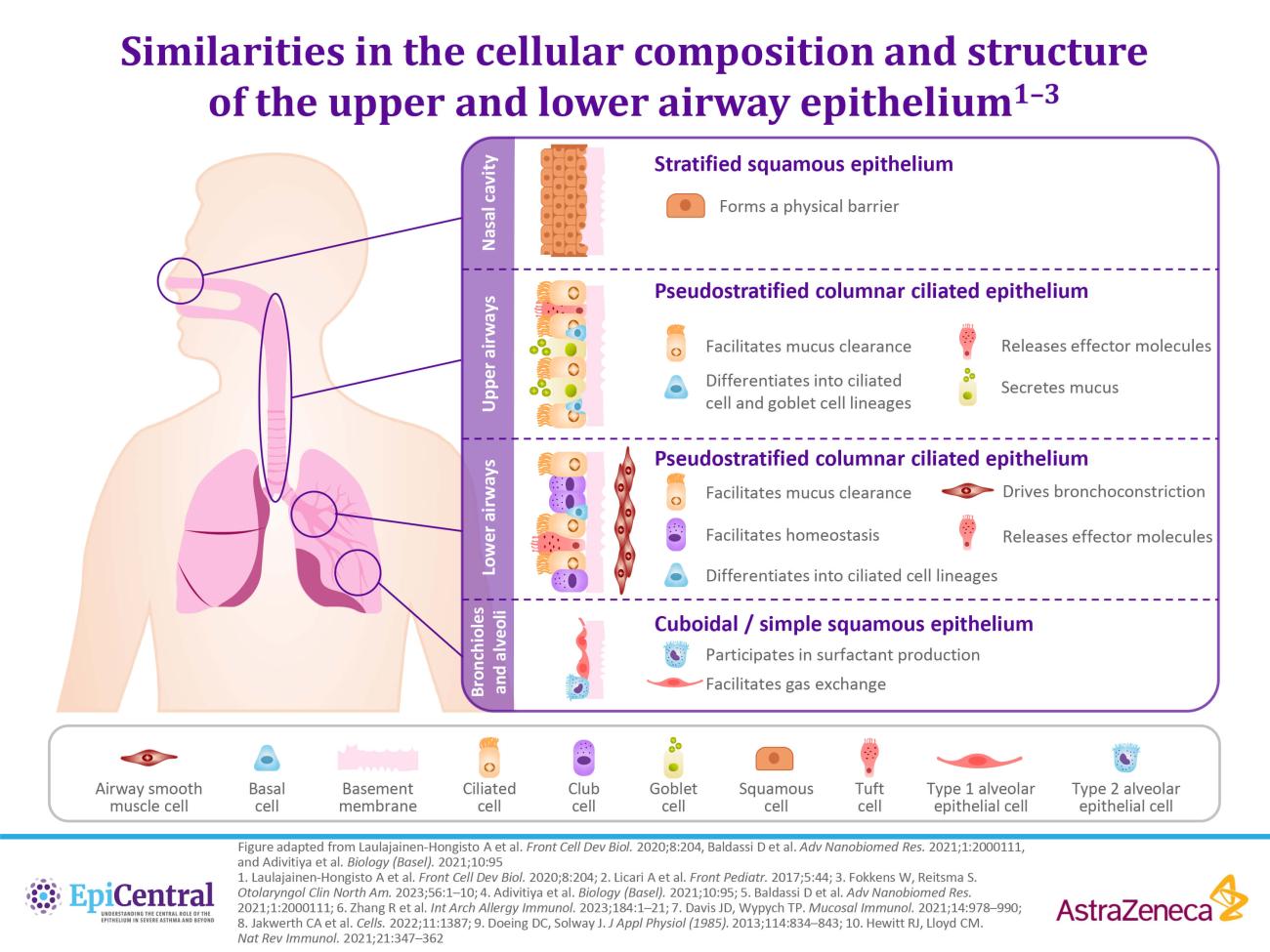
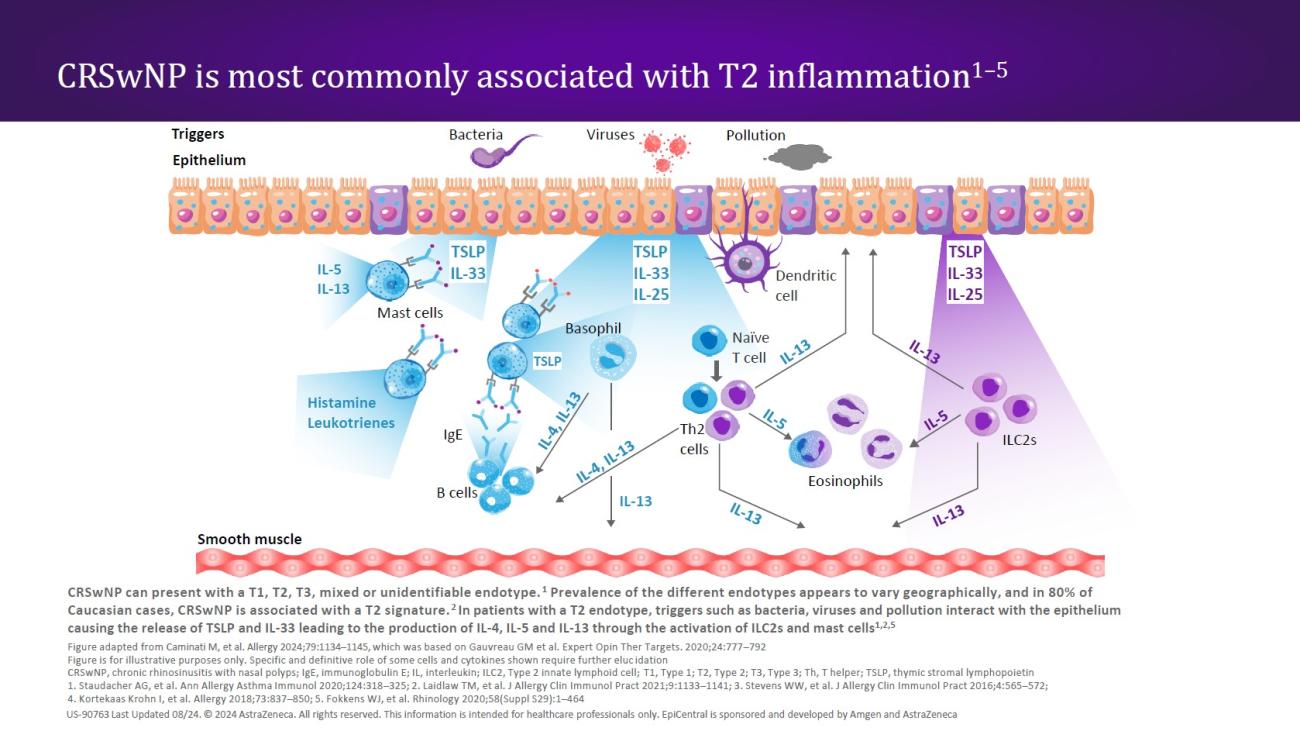
References
1. Roan F, et al. J Clin Invest. 2019;129:1441–1451;
2. Mitchell PD, O’Byrne PM. Chest. 2017;151:1338–1344;
3. Hellings PW, Steelant B. J Allergy Clin Immunol. 2020;145:1499–1509; 4. Fokkens W, Reitsma S. Otolaryngol Clin North Am. 2023;56:1–10; 5. Heijink IH, et al. Clin Exp Allergy. 2014;44:620–630; 6. Schleimer RP. Annu Rev Pathol. 2017;12:331–357; 7. Sharma K, et al. Cureus. 2022;14:e28501; 8. Jakwerth CA, et al. Cells. 2022;11:1387; 9. Stevens WW, et al. J Allergy Clin Immunol Pract. 2016;4:565–572; 10. Kicic A, et al. J Allergy Clin Immunol. 2020;145:1562–1573; 11. Yii ACA, et al. Allergy. 2018;73:1964–1978; 12. Licari A, et al. Front Pediatr. 2017;5:44; 13. Adivitiya, et al. Biology (Basel). 2021;10:95; 14. Zhang R, et al. Int Arch Allergy Immunol. 2023;184:1–21; 15. Laulajainen-Hongisto A, et al. Front Cell Dev Biol. 2020;8:204; 16. Bartemes KR, Kita H. Clin Immunol. 2012;143:222–235; 17. Fokkens WJ, et al. Rhinology. 2020;58(Suppl. S29):1–464; 18. Bousquet J, et al. Nat Rev Dis Primers. 2020;6:95; 19. Crystal RG, et al. Proc Am Thorac Soc. 2008;5:772–777; 20. Davis JD, Wypych TP. Mucosal Immunol. 2021;14:978–990; 21. Doeing DC, Solway J. J Appl Physiol (1985). 2013;114:834–843; 22. Orlandi RR, et al. Int Forum Allergy Rhinol. 2021;11:213–739; 23. Sedaghat AR, et al. J Allergy Clin Immunol Pract. 2022;10:1395–1403; 24. Claeys N, et al. Front Allergy. 2021;2:761388; 25. Wynne M, et al. Am J Rhinol Allergy. 2019;33:782–790; 26. Zhang M, et al. Int Immunopharmacol. 2023;121:110559; 27. Liao B, et al. Allergy. 2015;70:1169–1180; 28. Staudacher AG, et al. Ann Allergy Asthma Immunol. 2020;124:318–325; 29. Laidlaw TM, et al. J Allergy Clin Immunol Pract. 2021;9:1133–1141; 30. Sehmi R. Thorax. 2017;72:591–593; 31. Deng H, et al. J Asthma Allergy. 2021;14:839–850; 32. Liu R, et al. Front Immunol. 2021;12:530488; 33. Orlandi RR, et al. Int Forum Allergy Rhinol. 2016;6(Suppl. 1):S3–S21; 34. Cho SH, et al. J Allergy Clin Immunol Pract. 2016;4:575–582; 35. Bachert C, et al. J Asthma Allergy. 2021;14:127–134; 36. Head K, et al. Cochrane Database Syst Rev. 2016;4:CD011991; 37. Peters AT, et al. Allergy Asthma Proc. 2022;43:435–445; 38. Smith KA, et al. Int Forum Allergy Rhinol. 2019;9:402–408; 39. Starry A, et al. Allergy. 2022;77:2725–2736; 40. Mullol J, et al. J Allergy Clin Immunol Pract. 2022;10:1434–1453.e9; 41. Peters AT, et al. Ann Allergy Asthma Immunol. 2024;133(6 Suppl.):S8 (Abstract D020); 42. DeConde AS, et al. Laryngoscope. 2017;127:550–555; 43. van der Veen J, et al. Allergy. 2017;72:282–290; 44. Hwee J, et al. Allergy Asthma Clin Immunol. 2024;20:17; 45. Dykewicz MS, et al. J Allergy Clin Immunol. 2020;146:721–767; 46. Beard S. Prim Care. 2014;41:33–46; 47. Hellings PW, et al. Allergy. 2017;72:1657–1665; 48. Sin B, Togias A. Proc Am Thorac Soc. 2011;8:106–114; 49. Savouré M, et al. Clin Transl Allergy. 2022;12:e12130; 50. Wise SK, et al. Int Forum Allergy Rhinol. 2018;8:108–352; 51. Small P, et al. Allergy Asthma Clin Immunol. 2018;14(Suppl. 2):51; 52. Liu Y, et al. J Immunol Res. 2022;2022:4351345; 53. Shaaban R, et al. Am J Respir Crit Care Med. 2007;176:659–666; 54. Kaliner MA. World Allergy Organ J. 2009;2:98–101; 55. Greiwe JC, Bernstein JA. J Clin Med. 2019;8:2019; 56. Heffler E, et al. Clin Exp Allergy. 2018;48:1092–1106; 57. Shamil E, Hopkins C. Otolaryngol Clin North Am. 2023;56:157–168; 58. Miglani A, et al. Otolaryngol Clin North Am. 2023;56:11–22; 59. Caruso C, et al. Front Allergy. 2022;3:768408.