Developing the definition of remission in respiratory disease
The proposed definition of clinical remission in asthma and chronic rhinosinusitis with nasal polyps (CRSwNP) has evolved over time,1,2 with a definition previously focused on spontaneous or off-treatment remission, but has now evolved to focus on remission in patients who remain on treatment.3,4 Multiple criteria for remission in airway diseases have been proposed and it remains a clinically widely debated topic.1,2,5,6 These criteria have been developed based on definitions of remission in rheumatoid arthritis, ulcerative colitis, Crohn’s disease and systemic lupus erythematosus, where remission is a recognized clinical endpoint which has supported treatment advances and improved patient outcomes.1
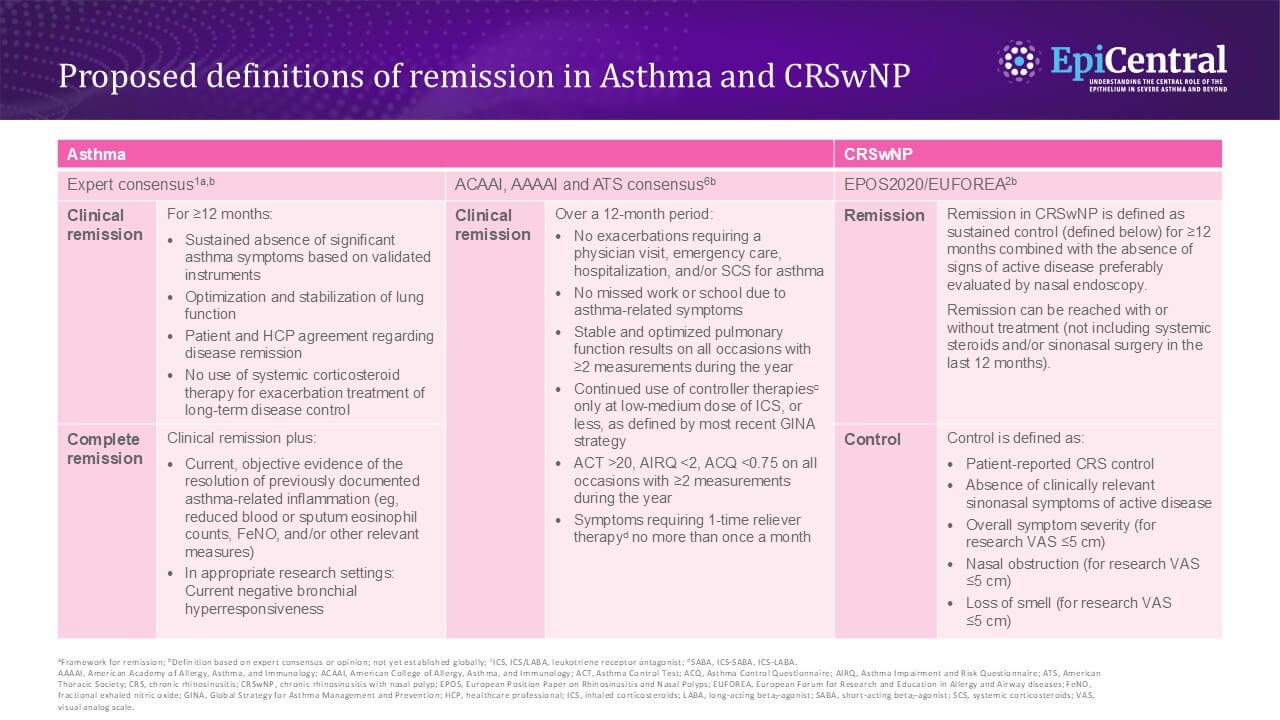
Broadly speaking, proposed definitions of remission in asthma include four components: no exacerbations, symptom control, no need for maintenance oral corticosteroids (OCS), and optimization and stabilization of lung function within a defined window.1,7 Similar criteria were proposed by a joint consensus of the American College of Allergy, Asthma, and Immunology, American Academy of Allergy, Asthma, and Immunology, and American Thoracic Society workgroup, but criteria for no missed work or school due to asthma-related symptoms were also included and restrictions around controller and reliever therapies were less stringent.6 In CRSwNP, remission is suggested to be defined as sustained control (patient-reported chronic rhinosinusitis (CRS) control, absence of clinically relevant sinonasal symptoms of active disease, lack of nasal obstruction, and no loss of smell) for ≥12 months.2 While remission seems to be less widely discussed in CRSwNP compared with asthma, this definition proposed by the European Forum for Research and Education in Allergy and Airway Diseases is being considered in the US and globally.8,9