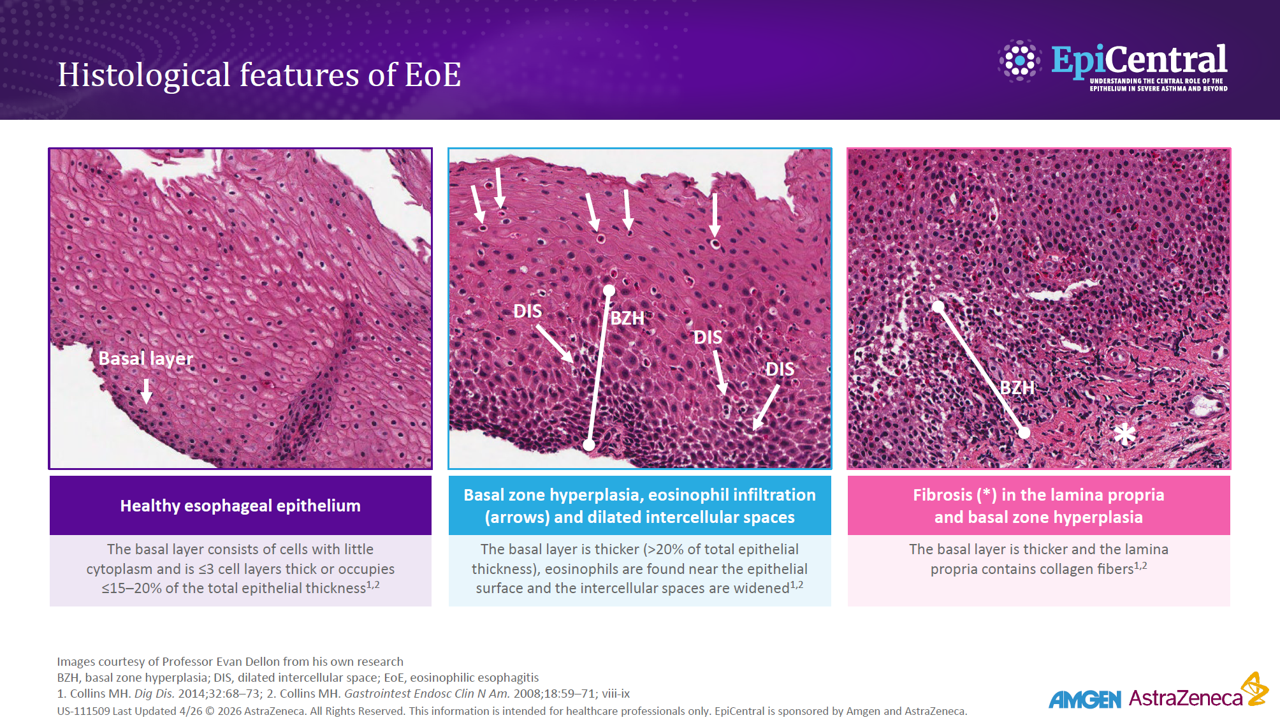
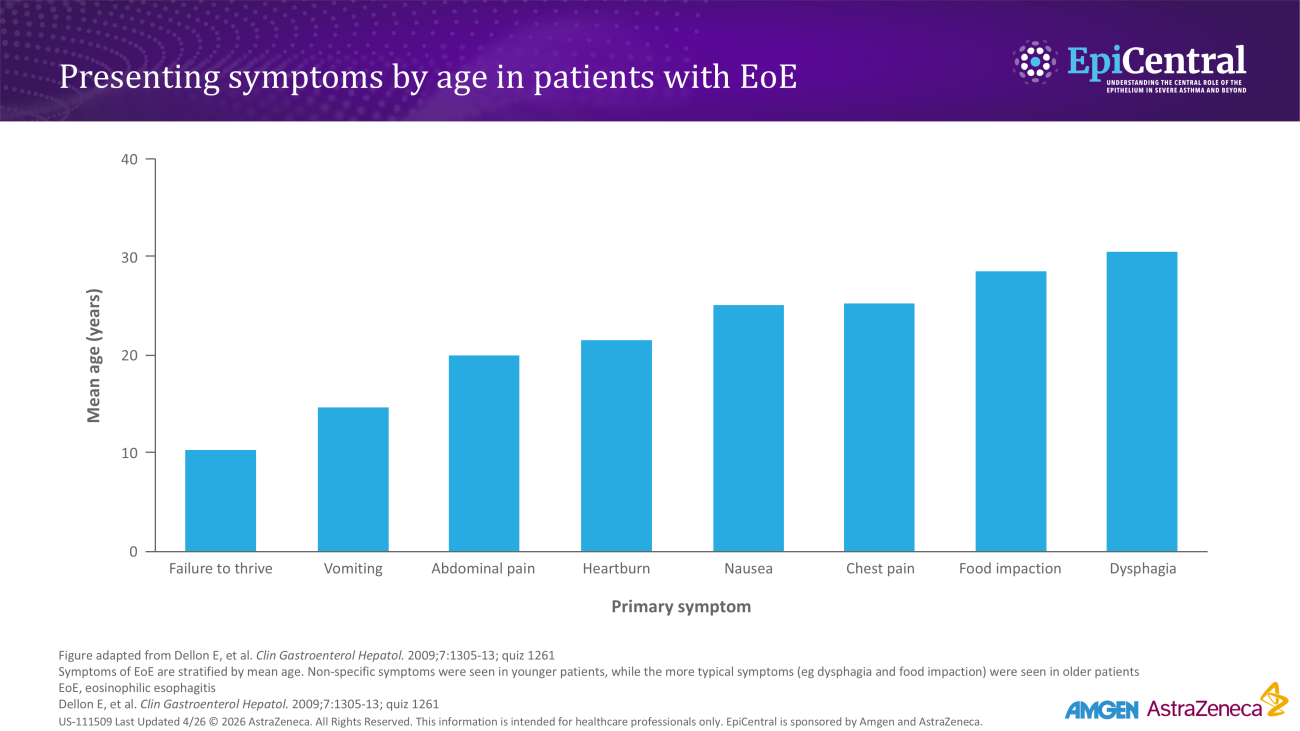
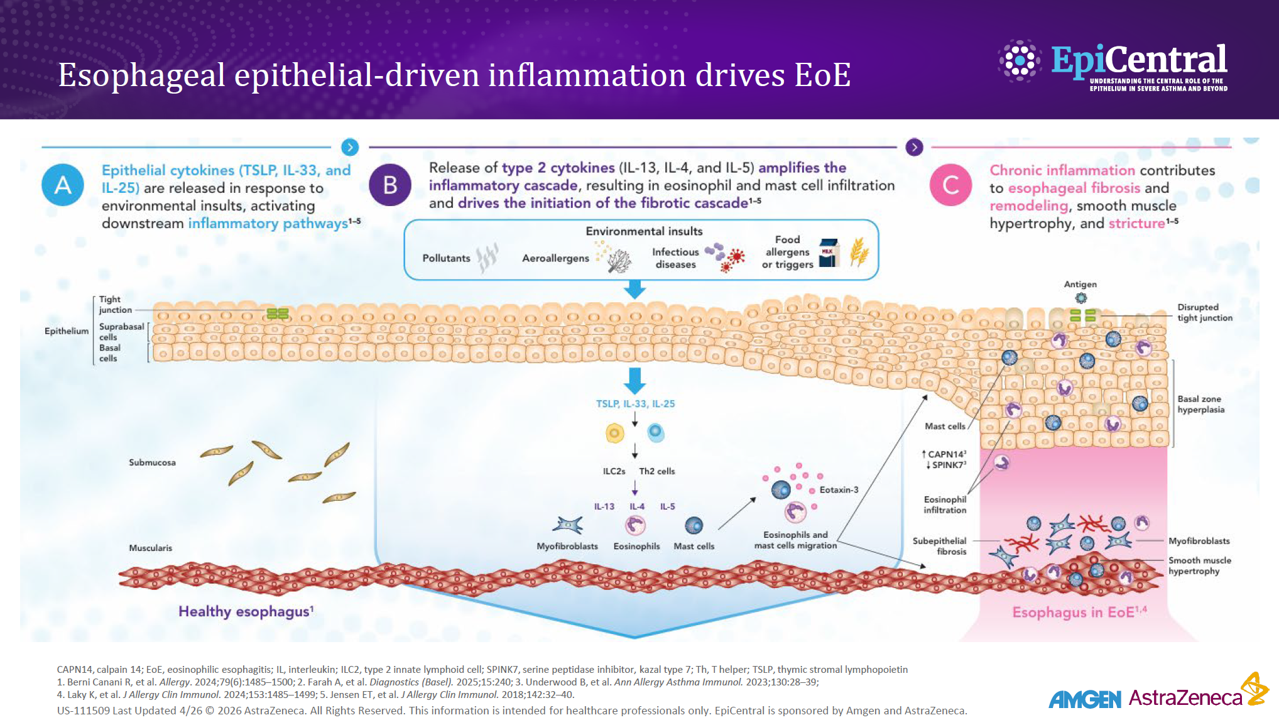
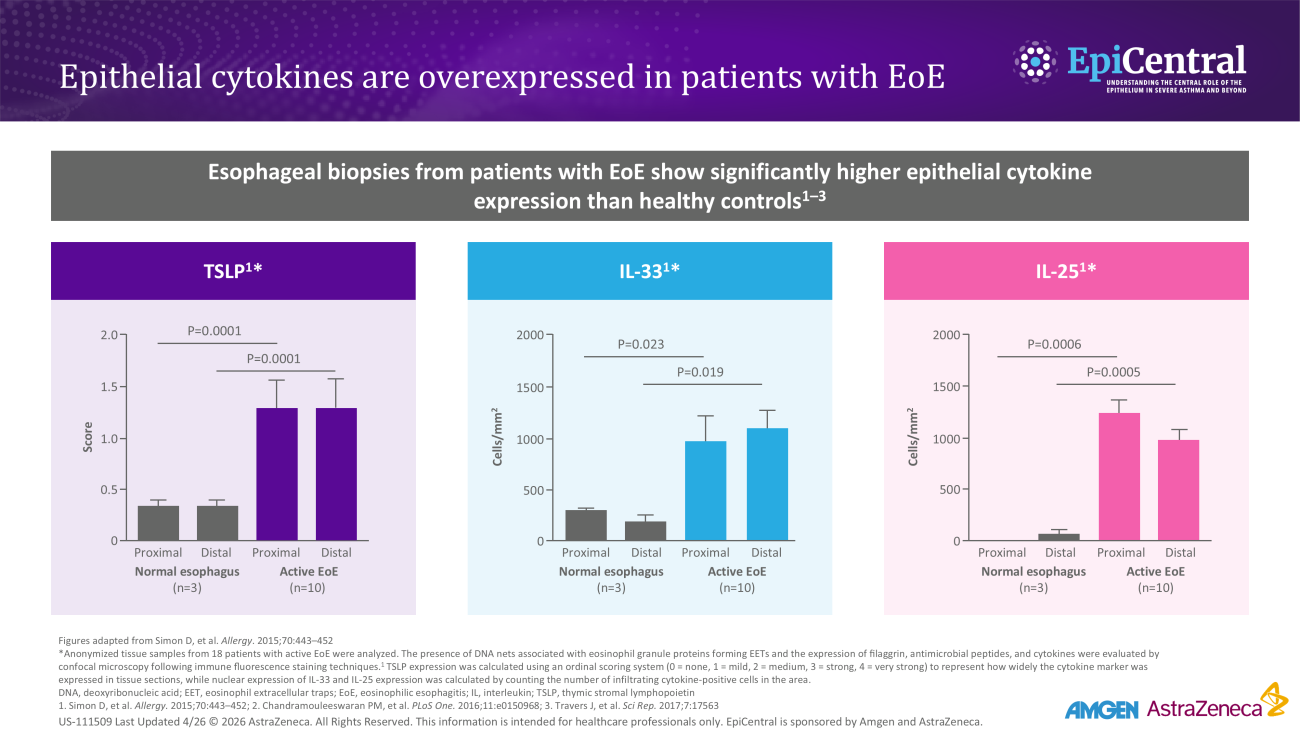
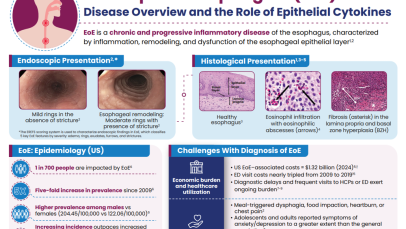
Rising prevalence of EoE in the USA
The incidence of EoE has increased rapidly in the USA in recent years.14 A recent study determined updated estimates of the prevalence and medical costs associated with EoE in the USA, using two large national insurance claim databases (MarketScan and Medicare).14 Healthcare utilization, including medications and endoscopic procedures, was quantified, and annual EoE-associated costs were estimated. Overall, 20,435 EoE cases in MarketScan (2022) and 1,913 EoE cases in Medicare (2017) were identified. Between 2009 and 2022, prevalence of EoE increased more than five-fold in the USA, from 28 cases/100,000 people to 163 cases/100,000 people.14 The economic impact is substantial, with total EoE-associated healthcare costs in the USA estimated to be $1.32 billion in 2024, when adjusted for inflation.14
Several risk factors contribute to EoE pathogenesis
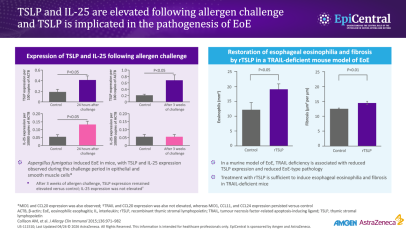
Ingested food allergens (including cow’s milk, wheat, soy, and eggs) are known triggers of epithelial dysfunction and T2 inflammation in EoE, and are considered to be risk factors and drivers of the disease.4,10,15 A disrupted epithelial barrier permits allergen penetration and subsequent immune activation.10 Although food allergens are known triggers of EoE, the mechanisms behind allergen-specific immune activation are not fully understood and further research is needed. Studies would suggest that the removal of specific foods, either by dietary elimination or use of hypoallergenic formulas, can lead to disease remission.15
The involvement of aeroallergens, infectious disease, and microbiome-altering factors in infancy (including antibiotic use during the first year of life, Cesarean delivery and pre-term birth, use of acid suppressants, or a stay in a neonatal intensive care unit), have been also been implicated in several studies.15,16 Although these factors are being investigated for a possible role in EoE, they have not yet been defined as proven risk factors for EoE.16
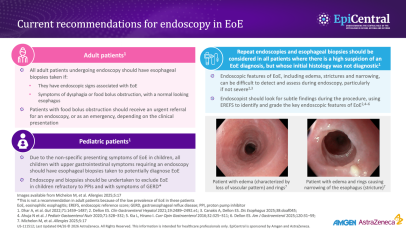
EoE is over five-times more likely to occur in people with atopy, and the more atopic comorbidities a patient has, the more likely they are to have EoE.17,18 Much like EoE, atopic diseases such as asthma, feature a disrupted epithelial barrier, which initiates and drives long-term T2 inflammation leading to clinical symptoms.4,19 Previous research suggests that between 60% and 80% of patients have concomitant allergic conditions, including IgE-mediated food allergies, asthma, atopic dermatitis, and allergic rhinitis.4 This highlights the importance of taking a detailed medical history when evaluating the patient in order to make the correct diagnosis and decisions about next steps for care.
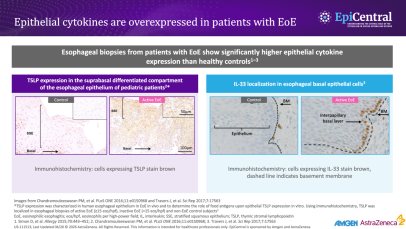
The importance of the epithelium and its interaction with the environment in disease pathogenesis has also been seen in children with EoE. A recent study assessed concurrent skin and esophageal dysfunction by evaluating ceramide levels in the skin of pediatric patients with EoE. Skin lipid composition of pediatric patients with EoE, but without atopic dermatitis (n=21), were compared with non-atopic dermatitis, non-EoE controls (n=17). It was found that the skin of patients with EoE have significant deficits in ceramide levels, particularly ultralong-chain fatty acid-containing ceramides, compared with the controls.20 These deficits may impair the skin’s barrier function and its interaction with environmental factors, such as allergens and microbes, supporting the concept that unified epithelial barrier dysfunction may contribute to both skin and esophageal atopic diseases.20